 Radioligands & Imaging Agents
Radioligands & Imaging Agents
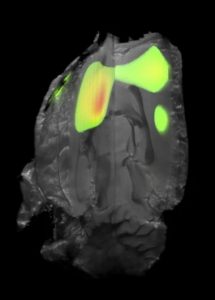
Our lab has pioneered the use of the agents below for non-invasive localization of chemogenetic and optogenetic switches in various species with the ultimate goal being human application.
[3H]ASEM – In vitro PSAM4-GlyR & PSAM4-5HT3 quantification [18F]ASEM – Longitudinal In vivo PSAM4-GlyR & PSAM4-5HT3 quantification [3H]Clozapine – In vitro hM3Dq/hM4Di quantification [3H]Compound 13 (C13) – In vitro hM3Dq/hM4Di quantification [18F]JHU37107 (J07) – Longitudinal In vivo hM3Dq/hM4Di quantificationOptogenetics (coming soon)
Related Publications
- Gomez, Bonaventura et al. 2017
- Magnus, Lee et al., 2019
- Bonaventura, Eldridge, Hu, et al., 2019
- Fleury Curado, et al., 2020
- Hu, Bonventura et al., 2020
 Actuators
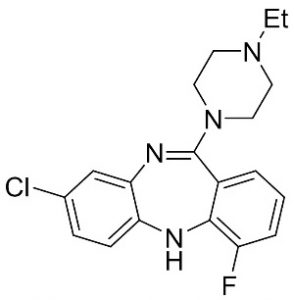
Actuators
Our lab has developed the first DREADD agonists that exhibit high affinity, high in vivo potency, and high brain penetrance in several species, which favors clinical translation. These compounds require very low systemic doses (<0.1 mg/kg) to facilitate rapid and remote activation of chemogenetic switches in the brain.
Related Publications
 Plasmids & Viral Vectors
Plasmids & Viral Vectors
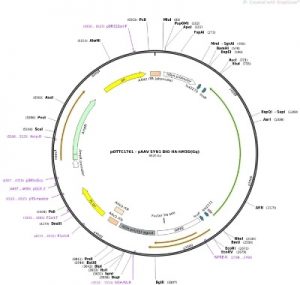
For translational and clinical gene therapy applications, chemogenetic/optogenetic switches need to be optimized to drive efficient expression and trafficking to the cell membrane, where their respective actuators would achieve maximum efficacy. For this reason, our goal has been to optimize existing chemogenetic/optogenetic gene therapy constructs for optimal expression and targeting. One way of doing this is to strip bulky and potentially toxic fluorescent reporters, typically used in such designs, and which are not useful for translational and clinical applications. Our strategy is to leverage the use of our translational PET-based reporters for non-invasive and longitudinal quantification of chemogenetic/optogenetic switches along with small epitopes (e.g. HA-tag) whenever in vitro detection would be necessary.
- pAAV SYN1 HA-hM3D(Gq)
- pAAV SYN1 HA-hM4D(Gi)
- pAAV SYN1 DIO HA-hM3D(Gq)
- pAAV SYN1 DIO HA-hM4D(Gi)
- pAAV EF1a HA-hM3D(Gq)
- pAAV EF1a HA-hM4D(Gi)
Related Publications
